Shunt Systems: This is a common treatment where a shunt (a thin tube) is implanted to divert excess cerebrospinal fluid from the brain to another part of the body where it can be absorbed. There may have been advancements in shunt technology to improve their effectiveness and reduce complications.
Endoscopic Third Ventriculostomy (ETV): This is a surgical procedure where a small hole is made in the floor of the third ventricle of the brain to allow excess cerebrospinal fluid to flow out and be absorbed. ETV is used in certain cases of non-communicating hydrocephalus.
Ventriculostomy with Catheter: Another surgical option involves inserting a catheter directly into the brain's ventricles to drain excess cerebrospinal fluid.
Intraventricular Endoscopy: Advances in minimally invasive techniques like endoscopy have allowed for more precise diagnosis and treatment of hydrocephalus.
US hydrocephalus shunts market companies are implementing strategies such as joint ventures and partnerships to extend their market shares. In 2020, Aesculap, Inc., partnered with Christoph Miethke GmbH & Co. KG (MIETHKE), and together they announced the launch of the M.blue hydrocephalus valve in the US. Its unique gravity technology is integrated with a fixed differential pressure unit in a valve, enabling a simple, position-dependent adjustment of the opening pressure during the patient’s most active time. Many market players are focused on developing “smart shunts” that could provide an understanding of flow or volume rates over time. Several industry partners are also developing devices that use thermodilution to estimate flow through a distal catheter to diagnose shunt malfunction. Thus, the increasing number of partnerships to aid in smart functioning of hydrocephalus shunts would lead to new future trends in the US hydrocephalus shunts market.
Researchers have been developing improved shunts materials to reduce the risk of blockages, infections, and other complications.
Antibacterial Coatings: Researchers have been investigating coatings for shunts that have antibacterial properties. These coatings can help reduce the risk of infection when the shunt is implanted.
Anti-Blockage Mechanisms: Some shunt designs incorporate features to prevent or mitigate blockages. For example, valves with anti-siphon mechanisms can help maintain proper flow and prevent over-drainage or blockages.
Magnetic Valve Adjustments: Some shunt systems use magnets to adjust valve settings non-invasively, reducing the need for repeated surgeries.
Biocompatible Materials: The use of biocompatible materials in shunt construction can minimize the risk of allergic reactions or other adverse responses from the body.
Remote Monitoring: Advancements in remote monitoring technology have allowed for tracking shunt performance and intracranial pressure, helping healthcare providers identify issues before they become severe.
Nanotechnology: Nanotechnology applications have been explored for creating shunt components that resist bacterial adhesion and improve the overall durability of the shunt.
For instance, in 2021, Penn State researchers have been actively working on a groundbreaking technology called HydroFix. This innovative surgical shunt system is specifically designed to address the numerous risks associated with existing shunt devices available in the market. Its primary aim is to offer a long-lasting treatment solution for patients with Normal Pressure Hydrocephalus (NPH). To advance this technology, the researchers established a startup called Cranial Devices Inc. They are currently in the process of seeking a license for the HydroFix technology from Penn State University.
Similarly, Rhaeos, Inc., a clinical-stage medical device company, is developing FlowSense, a patented technology and a noninvasive wireless, wearable skin patch. FlowSense is a wireless, noninvasive thermal flow sensor that is mounted on a patient’s neck overlying the shunt to detect the presence and magnitude of CSF. With FlowSense, shunt function can be monitored in clinics, in-patient settings, and emergency departments, thereby reducing unnecessary imaging, hospital length of stay, and readmission costs.
The US hydrocephalus shunts market growth is segmented into product, type, age group, and end-user. On the basis of product, the market is segmented into hydrocephalus valves, hydrocephalus catheters, neuronavigation systems, and others. The hydrocephalus valves is further segmented into adjustable pressure valves, and fix pressure valves. In terms of type, the US hydrocephalus shunts market is segmented into ventriculoperitoneal, ventriculoatrial, ventriculopleural, and lumboperitoneal. The ventriculoperitoneal segment led the market in 2022 and is expected to retain its dominance during the forecast period. A ventriculoperitoneal (VP) shunt reduces pressure exerted on the brain by fluid accumulation by diverting CSF from the brain's ventricles into the peritoneal cavity, the slot in the abdomen near digestive organs. Ventriculoperitoneal (VP) shunts are among the most commonly used tools for treating hydrocephalus. According to a research paper published in the National Center for Biotechnology Information (NCBI), a VP shunt has a high impact in advanced neurosurgical patient care. VP shunt can be lifesaving for benign disorders.
The US hydrocephalus shunts market, by age group, is segmented into pediatric and adults. The pediatric segment led the market in 2022 and is expected to retain its dominance during the forecast period by 2030. Hydrocephalus is the most common cause of brain surgery in children. A prenatal sonogram or MRI scan is recommended to identify hydrocephalus in a growing fetus. Infants generally show signs of progressive macrocephaly, whereas children older than 2 years show signs and symptoms of intracranial hypertension. Pediatric hydrocephalus can be dangerous if left untreated. Nevertheless, with early diagnosis and treatment, children often lead regular and healthy lives. According to the Hydrocephalus Association, 1–2 of every 1,000 babies have hydrocephalus in the US.
The US hydrocephalus shunts market, by end user, is segmented into hospitals, ambulatory surgical centers, others. The hospitals segment led the market in 2022 and is expected to retain its dominance during the forecast period by 2030. Hospitals provide healthcare to people through complicated but specialized scientific equipment. These facilities are classified into general, specialty, or government organizations. Hospitals serve a significant role by providing medical services to patient populations suffering from various diseases. The growth of the US hydrocephalus shunt market for the hospitals segment is attributed to an increase in the number of neurological surgeries, and a surge in demand for valves and neuronavigation systems in these facilities to treat the condition. Additionally, an increasing number of hospitals providing hydrocephalus surgeries in developed and developing countries is expected to boost the market growth in the future.
B. Braun SE, Sophysa SA, Integra LifeSciences Holdings Corp, KANEKA MEDIX CORP, Medtronic Plc, Natus Medical Inc, Anuncia Inc, Desu Medical are among the leading companies operating in the US hydrocephalus shunts market.
A few of the key primary and secondary sources referred to while preparing the report on the US
hydrocephalus shunts market are the World Health Organization (WHO), FDA (Food and Drug Administration), Hydrocephalus Association, National Institute of Neurological Disorders and Stroke (NINDS), University of California, Los Angeles Health Organization, National Center for Biotechnology Information, Children's Health Orange County, amongst others.
Strategic insights for the US Hydrocephalus Shunts provides data-driven analysis of the industry landscape, including current trends, key players, and regional nuances. These insights offer actionable recommendations, enabling readers to differentiate themselves from competitors by identifying untapped segments or developing unique value propositions. Leveraging data analytics, these insights help industry players anticipate the market shifts, whether investors, manufacturers, or other stakeholders. A future-oriented perspective is essential, helping stakeholders anticipate market shifts and position themselves for long-term success in this dynamic region. Ultimately, effective strategic insights empower readers to make informed decisions that drive profitability and achieve their business objectives within the market.
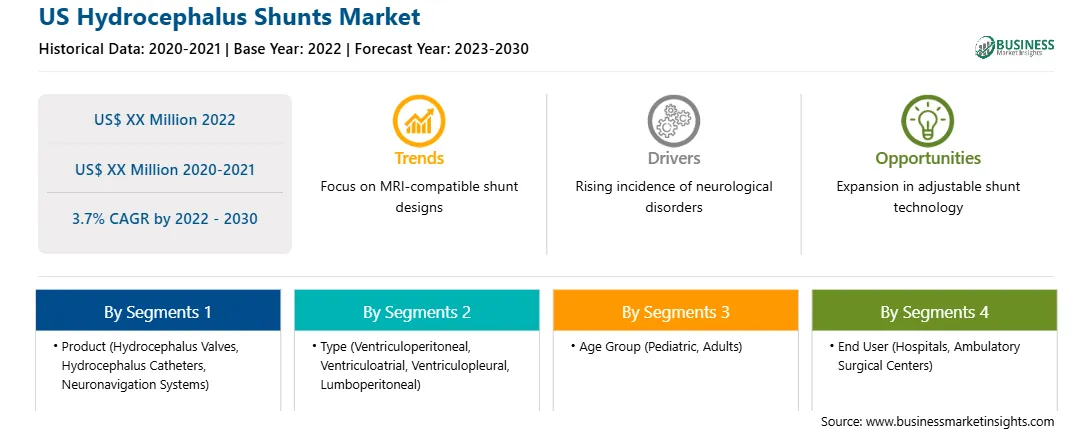
| Report Attribute | Details |
|---|---|
| Market size in 2022 | US$ XX Million |
| Market Size by 2030 | US$ XX Million |
| Global CAGR (2022 - 2030) | 3.7% |
| Historical Data | 2020-2021 |
| Forecast period | 2023-2030 |
| Segments Covered |
By Product
|
| Regions and Countries Covered | United States
|
| Market leaders and key company profiles |
The geographic scope of the US Hydrocephalus Shunts refers to the specific areas in which a business operates and competes. Understanding local distinctions, such as diverse consumer preferences (e.g., demand for specific plug types or battery backup durations), varying economic conditions, and regulatory environments, is crucial for tailoring strategies to specific markets. Businesses can expand their reach by identifying underserved areas or adapting their offerings to meet local demands. A clear market focus allows for more effective resource allocation, targeted marketing campaigns, and better positioning against local competitors, ultimately driving growth in those targeted areas.
The US Hydrocephalus Shunts Market is valued at US$ XX Million in 2022, it is projected to reach US$ XX Million by 2030.
As per our report US Hydrocephalus Shunts Market, the market size is valued at US$ XX Million in 2022, projecting it to reach US$ XX Million by 2030. This translates to a CAGR of approximately 3.7% during the forecast period.
The US Hydrocephalus Shunts Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the US Hydrocephalus Shunts Market report:
The US Hydrocephalus Shunts Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The US Hydrocephalus Shunts Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the US Hydrocephalus Shunts Market value chain can benefit from the information contained in a comprehensive market report.