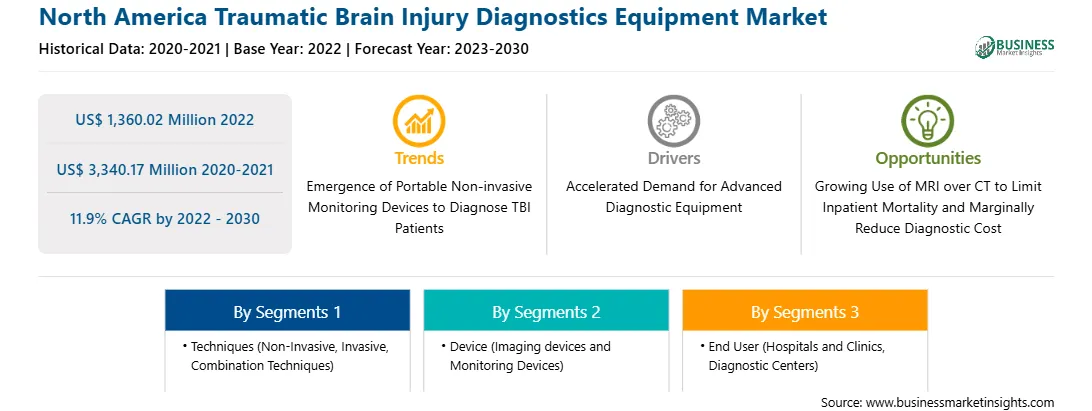
The North America traumatic brain injury diagnostics equipment market was valued at US$ 1,360.02 million in 2022 and is expected to reach US$ 3,340.17 million by 2030; it is estimated to register a CAGR of 11.9% from 2022 to 2030.
According to the Medical Research Council 2022 report, 10 million people across the world sustain traumatic brain injury (TBI) annually. Among these, the male population was 1.5 times higher than females admitted to hospitals for a head injury. As per The Economist Intelligence Unit report, the global healthcare burden of TBI is estimated to be around US$ 400 billion annually. The most common form of ABI is TBI due to an accident or stroke. The Centers for Disease Control and Prevention (CDC) report revealed that an estimated 1.7 million TBI-related emergency department visits, hospitalizations, and deaths occur annually in the US, especially among adults aged 75 years and older as they are at high risk of falling due to problems with gait and balance. Also, road accidents are the leading cause of TBI-related deaths in the US and are highest among adults aged 20–24 years. Therefore, manufacturers are developing innovative products to diagnose TBI.In October 2023, bioMérieux announced Conformité Européenne (CE) marking for "VIDAS TBI (GFAP, UCH-L1)," a test intended to improve the assessment of patients suffering from mild traumatic brain injury (mTBI). VIDAS TBI (GFAP, UCH-L1) test measures the concentration of glial fibrillary acidic protein (GFAP) and ubiquitin C-terminal hydrolase L1 (UCH-L1)—the two brain biomarkers released into the bloodstream starting from the first hour following a brain injury. It is an easy-to-interpret test providing a test window of up to 12 hours after injury, which can help in shortening total emergency department workup time.
The accelerated demand for advanced diagnostic equipment and quick and effective diagnosis for TBI patients are the key driving factors behind the market development. However, adverse effects of contrast/medium agent is hampering the market growth. The Centers for Disease Control and Prevention (CDC) report revealed that an estimated 2.5 million people suffer from TBI annually in the US. According to the KNAPP & ROBERTS report, 1 in every 6 Americans live with TBI-related disability in the US alone, accounting for approx. 5.3 million. With the rising prevalence of TBI, the economic cost accounts for US$ 76.5 billion. Among US$ 76.5 billion, US$ 11.5 billion accounts for direct medical costs and nearly US$ 65 billion for indirect costs. The leading causes of TBI include falls (45%), motor vehicle crashes (14.3%), assaults (10.7%), and unknown (19.0%). Further, companies are launching innovative products for the diagnosis of TBI. For instance, in August 2022, Abbott announced a new blood test for concussions to predict outcomes from brain injury and treatment interventions. The researchers used Abbott's "i-STAT TBI Plasma Test," the only FDA-cleared rapid test intended for concussion, and Abbott's core laboratory ARCHITECT instrument to measure two biomarkers in blood plasma associated with brain injury. The i-STAT TBI Plasma test measures the level of biomarkers released in the bloodstream in response to the brain injury; the level of biomarkers assist in determining the need for a CT scan. Additionally, Sense Neuro Diagnostics announced clearance to conduct clinical trials for hemorrhage detection. The new trial approved by the FDA Division of Neurosurgical, Neurointerventional, and Neurodiagnostic Devices began in June 2023, including up to 300 patients at 30 US, Canada, and India sites. This noninvasive technology has a potential to collect 360 data points within 2.5 seconds to detect brain hemorrhage or stroke type, thereby helping quick response by physicians, emergency department personnels, neuro ICU teams, and military field hospitals assessing and monitoring TBI.
Strategic insights for the North America Traumatic Brain Injury Diagnostics Equipment provides data-driven analysis of the industry landscape, including current trends, key players, and regional nuances. These insights offer actionable recommendations, enabling readers to differentiate themselves from competitors by identifying untapped segments or developing unique value propositions. Leveraging data analytics, these insights help industry players anticipate the market shifts, whether investors, manufacturers, or other stakeholders. A future-oriented perspective is essential, helping stakeholders anticipate market shifts and position themselves for long-term success in this dynamic region. Ultimately, effective strategic insights empower readers to make informed decisions that drive profitability and achieve their business objectives within the market.
| Report Attribute | Details |
|---|---|
| Market size in 2022 | US$ 1,360.02 Million |
| Market Size by 2030 | US$ 3,340.17 Million |
| Global CAGR (2022 - 2030) | 11.9% |
| Historical Data | 2020-2021 |
| Forecast period | 2023-2030 |
| Segments Covered |
By Techniques
|
| Regions and Countries Covered | North America
|
| Market leaders and key company profiles |
The geographic scope of the North America Traumatic Brain Injury Diagnostics Equipment refers to the specific areas in which a business operates and competes. Understanding local distinctions, such as diverse consumer preferences (e.g., demand for specific plug types or battery backup durations), varying economic conditions, and regulatory environments, is crucial for tailoring strategies to specific markets. Businesses can expand their reach by identifying underserved areas or adapting their offerings to meet local demands. A clear market focus allows for more effective resource allocation, targeted marketing campaigns, and better positioning against local competitors, ultimately driving growth in those targeted areas.
The North America traumatic brain injury diagnostics equipment market is categorized into techniques, device, end user, and country.
Based on techniques, the North America traumatic brain injury diagnostics equipment market is segmented into non-invasive, invasive, and combination techniques. The invasive segment held the largest North America traumatic brain injury diagnostics equipment market share in 2022. Furthermore, the non-invasive segment is subcategorized into electroencephalography (EEG), brain–computer interface (BCI), eye tracking and near-infrared spectroscopy (NIRS), magnetic resonance imaging (MRI), magnetoencephalography (MEG), transcranial magnetic stimulation (TMS), cerebral metabolic rate of oxygen (CMRO2), intracranial pressure, and others. Additionally, invasive segment is sub segmented into cerebral perfusion pressure (CPP) and pressure reactivity index (PRx), partial brain tissue oxygen pressure (PbtO2), cerebral microdialysis (CMD), intracranial pressure, and others.
In terms of device, the North America traumatic brain injury diagnostics equipment market is bifurcated into imaging devices and monitoring devices. The imaging devices segment held a larger North America traumatic brain injury diagnostics equipment market share in 2022.
By end user, the North America traumatic brain injury diagnostics equipment market is divided into hospitals and clinics, diagnostic centers, and others. The hospitals and clinics segment held the largest North America traumatic brain injury diagnostics equipment market share in 2022.
By country, the North America traumatic brain injury diagnostics equipment market is segmented into the US, Canada, and Mexico. The US dominated the North America traumatic brain injury diagnostics equipment market share in 2022.
GE HealthCare Technologies Inc, Elekta AB, Integra LifeSciences Holdings Corp, Natus Medical Inc, Raumedic AG, BrainScope Co Inc, Soterix Medical Inc, Medtronic Plc, Vivonics Inc, NanoDx Inc, Compumedics Ltd, Sense Diagnostics Inc, NeuraSignal Inc, Neurovigil Inc, and Luciole Medical AG, are some of the leading companies operating in the North America traumatic brain injury diagnostics equipment market.
The North America Traumatic Brain Injury Diagnostics Equipment Market is valued at US$ 1,360.02 Million in 2022, it is projected to reach US$ 3,340.17 Million by 2030.
As per our report North America Traumatic Brain Injury Diagnostics Equipment Market, the market size is valued at US$ 1,360.02 Million in 2022, projecting it to reach US$ 3,340.17 Million by 2030. This translates to a CAGR of approximately 11.9% during the forecast period.
The North America Traumatic Brain Injury Diagnostics Equipment Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the North America Traumatic Brain Injury Diagnostics Equipment Market report:
The North America Traumatic Brain Injury Diagnostics Equipment Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The North America Traumatic Brain Injury Diagnostics Equipment Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the North America Traumatic Brain Injury Diagnostics Equipment Market value chain can benefit from the information contained in a comprehensive market report.