Cell and gene therapy manufacturing is a complex process, which makes the proper execution and overseeing of the operation crucial. Cell and gene therapy manufacturers have a limited number of qualified personnel who know biological and process engineering. Moreover, for experienced teams, managing the attempts to reach the first clinical trial using a manual, and open manufacturing method and then building a more commercially suitable process can be tricky. Therefore, these enterprises choose to work with contract development and manufacturing organizations (CDMOs) to accelerate their clinical studies and commercialization process. CDMOs provide product development, manufacturing, clinical trial support, and commercialization services to cell and gene therapy companies on a contract basis. Partnering with a CDMO enables scalability, speed to market, access to technical expertise without overhead costs, and cost efficiencies for cell and gene therapy manufacturers. In April 2022, ThermoGenesis established a CDMO facility in California, US to provide CDMO services to cell and gene therapy manufacturers, using its expertise in T-cell receptor (TCR), chimeric antigen receptor-T cell (CAR-T cell), tumor-infiltrating leukocyte (TIL), iPSC, natural killer cell (NK), and mesenchymal stem cell (MSC) manufacturing. Outsourcing cell and gene therapy manufacturing to CDMOs proves cost-effective for manufacturers. Thus, the increasing preference for outsourcing growing cell and gene therapy manufacturing to CDMOs fuels the North America cell and gene therapy manufacturing services market growth.
Cell and gene therapies (CGTs) treat patients suffering from serious and rare diseases with unaddressed therapeutic needs. Manufacturing CGTs is a highly complex process, with the insufficiency of infrastructure and expertise being a major limiting factor. Logistical challenges associated with intermediates and the final product also limit the CGT manufacturing capacity of companies. The CGT manufacturing process involves the extraction of autologous cells through "apheresis," dispatching them to specialized labs, and sending them back to clinics for administration into patients, all of which must be performed with strict quality control. The US Food and Drug Administration (USFDA) has approved only 7 CGT drugs, with the pipeline of new products reaching ~1,200 experimental therapies. Half of these are in Phase 2 clinical trials, with estimates of annual sales growth accounting for 15% for cell therapies and ~30% for gene therapies, as per the estimates of the Chemical & Engineering News report 2022. On March 31, 2022, CELL Technologies Inc. announced the submission of the clinical data of its stem cell program in pain and arthritis for approval by Health Canada to help patients access evidence-based and regulatory-approved stem cell procedures across Canada. Thus, the abovementioned factors are expected to promote the cell and gene therapy manufacturing services market growth during the forecast period. Thus, the abovementioned factors are responsible for the growth of the North America cell and gene therapy manufacturing services market.
The North America cell and gene therapy manufacturing services market is segmented into type, indication, application, end user, and country.
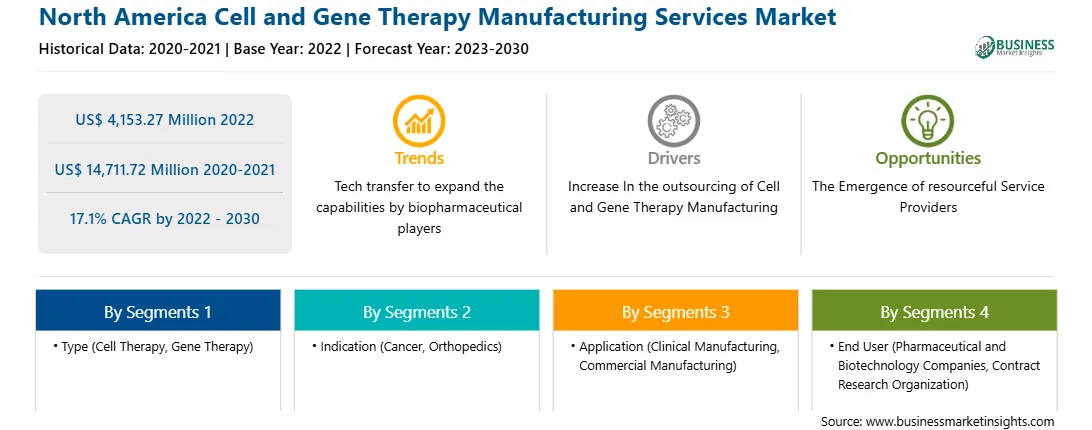
Based on type, the North America cell and gene therapy manufacturing services market is bifurcated into cell therapy and gene therapy. In 2022, the cell therapy segment registered a larger share in the North America cell and gene therapy manufacturing services market. The cell therapy segment is further segmented into autologous and allogenic. The gene therapy segment is further segmented into viral and non-viral vector.
Based on indication, the North America cell and gene therapy manufacturing services market is segmented into cancer, orthopedics, and others. In 2022, the cancer segment registered the largest share in the North America cell and gene therapy manufacturing services market.
Based on application, the North America cell and gene therapy manufacturing services market is segmented into clinical manufacturing and commercial manufacturing. In 2022, the commercial manufacturing segment registered the largest share in the North America cell and gene therapy manufacturing services market.
Based on end user, the North America cell and gene therapy manufacturing services market is bifurcated into pharmaceutical and biotechnology companies and contract research organization (CROs). In 2022, the pharmaceutical and biotechnology companies segment registered a larger share in the North America cell and gene therapy manufacturing services market.
Based on country, the North America cell and gene therapy manufacturing services market is segmented into the US, Canada, Mexico. In 2022, the US registered the largest share in the North America cell and gene therapy manufacturing services market.
Catalent Inc, Charles River Laboratories International Inc, FUJIFILM Holdings Corp, Lonza Group AG, Merck KgaA, National Resilience Inc, Nikon Corp, Oxford BioMedica Plc, Takara Bio Inc, Thermo Fisher Scientific Inc, and WuXi AppTec Co Ltd are some of the leading companies operating in the North America cell and gene therapy manufacturing services market.
Strategic insights for the North America Cell and Gene Therapy Manufacturing Services provides data-driven analysis of the industry landscape, including current trends, key players, and regional nuances. These insights offer actionable recommendations, enabling readers to differentiate themselves from competitors by identifying untapped segments or developing unique value propositions. Leveraging data analytics, these insights help industry players anticipate the market shifts, whether investors, manufacturers, or other stakeholders. A future-oriented perspective is essential, helping stakeholders anticipate market shifts and position themselves for long-term success in this dynamic region. Ultimately, effective strategic insights empower readers to make informed decisions that drive profitability and achieve their business objectives within the market.
| Report Attribute | Details |
|---|---|
| Market size in 2022 | US$ 4,153.27 Million |
| Market Size by 2030 | US$ 14,711.72 Million |
| Global CAGR (2022 - 2030) | 17.1% |
| Historical Data | 2020-2021 |
| Forecast period | 2023-2030 |
| Segments Covered |
By Type
|
| Regions and Countries Covered | North America
|
| Market leaders and key company profiles |
The geographic scope of the North America Cell and Gene Therapy Manufacturing Services refers to the specific areas in which a business operates and competes. Understanding local distinctions, such as diverse consumer preferences (e.g., demand for specific plug types or battery backup durations), varying economic conditions, and regulatory environments, is crucial for tailoring strategies to specific markets. Businesses can expand their reach by identifying underserved areas or adapting their offerings to meet local demands. A clear market focus allows for more effective resource allocation, targeted marketing campaigns, and better positioning against local competitors, ultimately driving growth in those targeted areas.
1. Catalent Inc
2. Charles River Laboratories International Inc
3. FUJIFILM Holdings Corp
4. Lonza Group AG
5. Merck KgaA
6. National Resilience Inc
7. Nikon Corp
8. Oxford BioMedica Plc
9. Takara Bio Inc
10. Thermo Fisher Scientific Inc
11. WuXi AppTec Co Ltd?
The North America Cell and Gene Therapy Manufacturing Services Market is valued at US$ 4,153.27 Million in 2022, it is projected to reach US$ 14,711.72 Million by 2030.
As per our report North America Cell and Gene Therapy Manufacturing Services Market, the market size is valued at US$ 4,153.27 Million in 2022, projecting it to reach US$ 14,711.72 Million by 2030. This translates to a CAGR of approximately 17.1% during the forecast period.
The North America Cell and Gene Therapy Manufacturing Services Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the North America Cell and Gene Therapy Manufacturing Services Market report:
The North America Cell and Gene Therapy Manufacturing Services Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The North America Cell and Gene Therapy Manufacturing Services Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the North America Cell and Gene Therapy Manufacturing Services Market value chain can benefit from the information contained in a comprehensive market report.