Increasing Clinical Trials and Rising GMP Demand to Fuel Middle East & Africa GMP Cell Therapy
Consumables Market in Coming Years
Cell therapies are constantly evolving, improving, and providing new treatment options for various disorders, such as cancer, autoimmune disorders, and neurological disorders. Immunotherapy is a new approach to cancer treatment. Cell therapies are currently being evaluated in clinical trials for various cancer types, separately and combined with other treatments. Additionally, the cellular immunotherapy targets and new immunotherapy approaches are investigated through clinical trials. Since 2017, FDA has approved approximately six chimeric antigen receptor (CAR) T-cell therapies for the treatment of blood cancer, including lymphomas, some leukemia forms, and multiple myeloma. More than 500 CAR-T and 17 chimeric antigen receptor natural killer (CAR-NK) cell trials are currently being conducted across the world, including the four CAR-T cell products—Yescarta, Tecartus, Kymriah, and Breyanzi—that are already available in the market. Further, the CAR cellular therapies field is newly developed, and preclinical and clinical studies have already shown remarkable results. It is believed that CAR-based cellular therapies are profoundly promising and will lead the development of approved personalized therapeutic options in the coming years. With new therapeutic modalities emerging in the cell and gene space (CAR-T), companies are required to develop, evaluate, and test these therapies in clinical trials, along with maintaining patient safety. These emerging therapies require a scientific standard of controls and GMPs to ensure patient safety. Thus, it is important to ensure that the equipment used during these processes is compatible with GMP manufacturing environments for cell therapies and that all clinical consumables are manufactured under conditions that fulfill GMP requirements appropriate for the phase and scale of manufacturing. Therefore, the increasing clinical trials in various fields will raise the demand for GMP cell therapy consumables in the coming years, thereby driving the Middle East & Africa GMP cell therapy consumables market.
Middle East & Africa GMP Cell Therapy Consumables Market Overview
The UAE, Saudi Arabia, and South Africa are the three major countries in the Middle East & Africa. Saudi Arabia was the largest market for GMP cell therapy consumables in this region in 2021. The Middle East & Africa GMP cell therapy consumables market is anticipated to grow at a CAGR of 25.6% during the forecast period; this growth is attributed to the rising healthcare expenditure and increasing government initiatives in funding and healthcare-related policies. The UAE is a developed country in the Gulf Cooperation Council (GCC) that is focused on creating world-class health facilities. The healthcare facilities in the UAE expanded significantly in the last few years as the UAE government is collaborating with US-based companies and healthcare facilities, improving healthcare facilities, services, and patient care by providing cutting-edge technologies. The UAE government mostly funds the country's healthcare system. According to the International Trade Administration US Department of Commerce, healthcare spending in UAE is forecasted to rise to US$ 18.3 billion by 2023. Additionally, the Dubai International Pharmaceuticals & Technologies Conference & Exhibition (DUPHAT) reported that medical tourism is expanding in the UAE. The medical tourism industry in the UAE generated AED 12.1 billion (US$ 3.29 billion) in 2018, witnessing a 5.5% annual growth rate. The country recorded ~350,118 visitors, registering a rise of 4% in health tourism arrivals in 2019 from the previous year. It is anticipated that this will rise to 500,000 medical tourists per year. Further, the growing population in the country is prone to various infections and diseases, including cancer, creating immense demand for cell-based therapies. Thus, the increasing medical tourism, rising government expenditure, and growing adoption of cell therapies will increase the demand for GMP cell therapy consumables and propel the growth of the GMP cell therapy consumables market in the UAE during the forecast period.
Strategic insights for the Middle East & Africa GMP Cell Therapy Consumables provides data-driven analysis of the industry landscape, including current trends, key players, and regional nuances. These insights offer actionable recommendations, enabling readers to differentiate themselves from competitors by identifying untapped segments or developing unique value propositions. Leveraging data analytics, these insights help industry players anticipate the market shifts, whether investors, manufacturers, or other stakeholders. A future-oriented perspective is essential, helping stakeholders anticipate market shifts and position themselves for long-term success in this dynamic region. Ultimately, effective strategic insights empower readers to make informed decisions that drive profitability and achieve their business objectives within the market. The geographic scope of the Middle East & Africa GMP Cell Therapy Consumables refers to the specific areas in which a business operates and competes. Understanding local distinctions, such as diverse consumer preferences (e.g., demand for specific plug types or battery backup durations), varying economic conditions, and regulatory environments, is crucial for tailoring strategies to specific markets. Businesses can expand their reach by identifying underserved areas or adapting their offerings to meet local demands. A clear market focus allows for more effective resource allocation, targeted marketing campaigns, and better positioning against local competitors, ultimately driving growth in those targeted areas.Middle East & Africa GMP Cell Therapy Consumables Strategic Insights
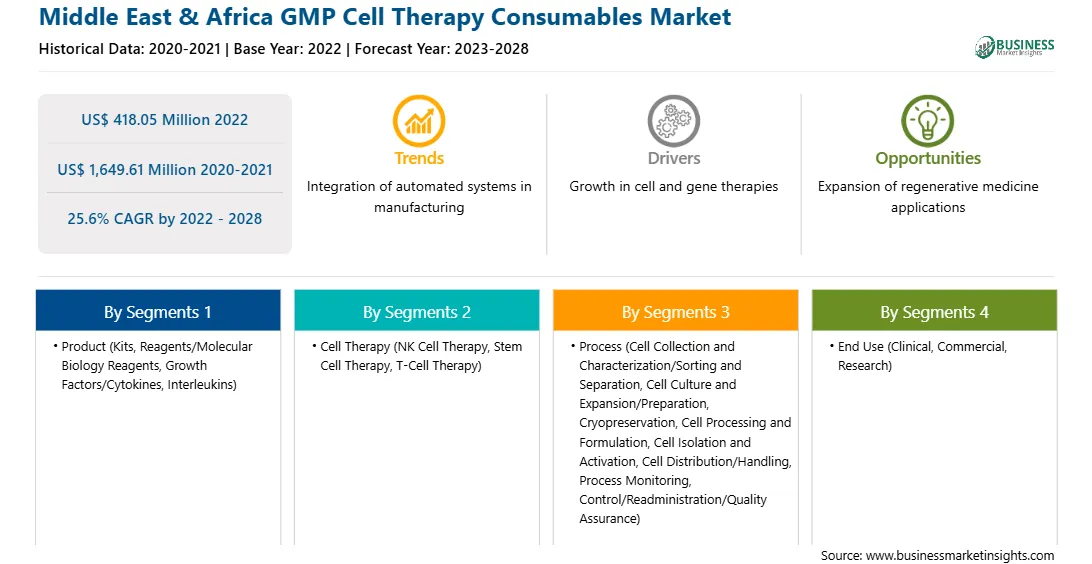
Middle East & Africa GMP Cell Therapy Consumables Report Scope
Report Attribute
Details
Market size in 2022
US$ 418.05 Million
Market Size by 2028
US$ 1,649.61 Million
Global CAGR (2022 - 2028)
25.6%
Historical Data
2020-2021
Forecast period
2023-2028
Segments Covered
By Product
By Cell Therapy
By Process
By End Use
Regions and Countries Covered
Middle East and Africa
Market leaders and key company profiles
Middle East & Africa GMP Cell Therapy Consumables Regional Insights
Middle East & Africa GMP Cell Therapy Consumables Market Segmentation
The Middle East & Africa GMP cell therapy consumables market is segmented based on product, cell therapy, process, end use, and country.
Based on product, the Middle East & Africa GMP cell therapy consumables market is segmented into kits, reagents/molecular biology reagents, growth factors/cytokines and interleukins (including protein and nucleic acid purification buffers), and others. The kits segment held the largest share of the Middle East & Africa GMP cell therapy consumables market in 2022.
Based on cell therapy, the Middle East & Africa GMP cell therapy consumables market is segmented into NK cell therapy, stem cell therapy, T-cell therapy, and others. The T-cell therapy segment registered the largest share of the Middle East & Africa GMP cell therapy consumables market in 2022.
Based on process, the Middle East & Africa GMP cell therapy consumables market is segmented into cell collection and characterization/sorting and separation, cell culture and expansion/preparation, cryopreservation, cell processing and formulation, cell isolation and activation, cell distribution/handling, process monitoring and control/readministration/quality assurance, and others. The cell collection and characterization/sorting and separation segment held the largest share of the Middle East & Africa GMP cell therapy consumables market in 2022.
Based on end use, the Middle East & Africa GMP cell therapy consumables market is segmented into clinical, commercial, and research. The clinical segment held the largest share of the Middle East & Africa GMP cell therapy consumables market in 2022.
Based on country, the Middle East & Africa GMP cell therapy consumables market is segmented into Saudi Arabia, South Africa, the UAE, and the Rest of Middle East & Africa. Saudi Arabia dominated the Middle East & Africa GMP cell therapy consumables market in 2022.
Bio-Techne Corp; BPS Bioscience Inc; Corning Inc; FUJIFILM Irvine Scientific Inc; Global Life Sciences Solutions USA LLC; Lonza Group AG; Merck KGaA; Miltenyi Biotec BV & Co KG; Sartorius AG; and Thermo Fisher Scientific Inc are the leading companies operating in Middle East & Africa GMP cell therapy consumables market.
The Middle East & Africa GMP Cell Therapy Consumables Market is valued at US$ 418.05 Million in 2022, it is projected to reach US$ 1,649.61 Million by 2028.
As per our report Middle East & Africa GMP Cell Therapy Consumables Market, the market size is valued at US$ 418.05 Million in 2022, projecting it to reach US$ 1,649.61 Million by 2028. This translates to a CAGR of approximately 25.6% during the forecast period.
The Middle East & Africa GMP Cell Therapy Consumables Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Middle East & Africa GMP Cell Therapy Consumables Market report:
The Middle East & Africa GMP Cell Therapy Consumables Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Middle East & Africa GMP Cell Therapy Consumables Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Middle East & Africa GMP Cell Therapy Consumables Market value chain can benefit from the information contained in a comprehensive market report.