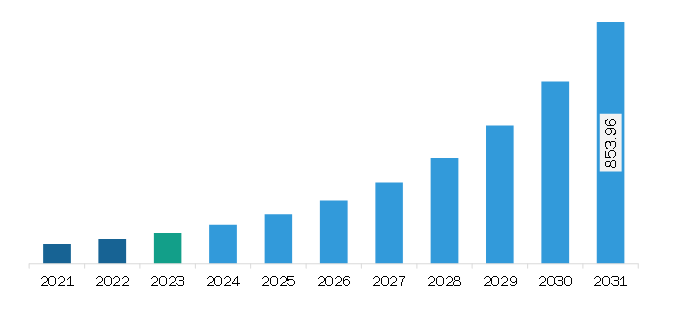
The Europe medical exoskeleton market was valued at US$ 107.48 million in 2023 and is expected to reach US$ 853.96 million by 2031; it is estimated to register a CAGR of 29.6% from 2023 to 2031.
Despite the advancements in the medical industry, stroke continues to be a major public health issue. Stroke ranks second in the world in terms of causes of mortality and is the main cause of disability. According to the Global Stroke Factsheet 2022, the lifetime risk of having a stroke has risen by 50% in the past 17 years, with an estimated 1 in 4 people expected to experience one at some point in their lives. Between 1990 and 2019, there was a rise of 70% in the incidence of stroke, 43% in the number of stroke-related deaths, 102% in the frequency of stroke, and 143% in Disability Adjusted Life Years (DALY). The most notable aspect is that lower- and lower-middle-income countries account for the majority of the world's stroke burden (86% of stroke-related fatalities and 89% of DALYs). In the European Union, there were 1.12 million stroke incidents, 9.53 million stroke survivors, 0.46 million stroke-related deaths, and 7.06 million years of life lost due to disability from stroke in 2017. Further, the number of people living with stroke is predicted to rise by 27% between 2017 and 2047, primarily as a result of aging populations and higher survival rates. It is anticipated that differences will continue to exist between nations, indicating potential for advancements in case management and prevention, especially in Eastern Europe. Therefore, the increasing incidences of stroke have boosted the demand for exoskeleton systems for rehabilitation of stroke patients to help improve the quality of life.
In addition, musculoskeletal disorders impact the mobility of an individual. The increasing prevalence of musculoskeletal disorders, such as tendinitis, osteopenia, sarcopenia, and low back injuries, result in a growing need for exoskeleton robotic systems. According to the World Health Organization (WHO), ~1.71 billion people globally are affected by musculoskeletal disease, and lower back pain is a leading cause of disability in 160 countries. These exoskeletons assist patients in managing their pain, as well as help regain their mobility, thereby improving their overall quality of life. Thus, the increasing prevalence of stroke and musculoskeletal disorders has led to the use of exoskeleton robotic systems for rehabilitation.
The European medical exoskeleton market is segmented into Germany, the UK, France, Italy, Spain, and the Rest of Europe. The region holds a significant share of the global medical exoskeleton market and is expected to register a notable CAGR. Market players in the country are adopting inorganic growth strategies such as partnerships and collaborations for market expansion and growth. In August 2022, German Bionic (a global technology leader in the development of smart power suits) and Mubea (the top German automotive supplier) entered a cooperation agreement. By assisting the world's top developer of robotic exoskeletons in production, Mubea tapped into a new and quite promising future sector. Similarly, in April 2020, ReWalk Robotics Ltd. announced the national agreement with top German Statutory Health Insurers ("SHI"), Deutsche Angestellten-Krankenkasse – Gesundheit ("DAK-Gesundheit"), and Techniker Krankenkasse ("TK"). This agreement can permit any eligible beneficiary with a spinal cord injury to get a ReWalk 6.0 exoskeleton system. TK and DAK, representing 10.6 million and 5.6 million beneficiaries in Germany, respectively, are two of the largest SHIs in the country; these agreements are expected to help set the standard for exoskeleton procurement in Germany.
Strategic insights for the Europe Medical Exoskeleton provides data-driven analysis of the industry landscape, including current trends, key players, and regional nuances. These insights offer actionable recommendations, enabling readers to differentiate themselves from competitors by identifying untapped segments or developing unique value propositions. Leveraging data analytics, these insights help industry players anticipate the market shifts, whether investors, manufacturers, or other stakeholders. A future-oriented perspective is essential, helping stakeholders anticipate market shifts and position themselves for long-term success in this dynamic region. Ultimately, effective strategic insights empower readers to make informed decisions that drive profitability and achieve their business objectives within the market.

| Report Attribute | Details |
|---|---|
| Market size in 2023 | US$ 107.48 Million |
| Market Size by 2031 | US$ 853.96 Million |
| Global CAGR (2023 - 2031) | 29.6% |
| Historical Data | 2020-2021 |
| Forecast period | 2023-2031 |
| Regions and Countries Covered | Europe
|
| Market leaders and key company profiles |
The geographic scope of the Europe Medical Exoskeleton refers to the specific areas in which a business operates and competes. Understanding local distinctions, such as diverse consumer preferences (e.g., demand for specific plug types or battery backup durations), varying economic conditions, and regulatory environments, is crucial for tailoring strategies to specific markets. Businesses can expand their reach by identifying underserved areas or adapting their offerings to meet local demands. A clear market focus allows for more effective resource allocation, targeted marketing campaigns, and better positioning against local competitors, ultimately driving growth in those targeted areas.
The Europe medical exoskeleton market is categorized into component, type, extremity, application, mobility, end users, and country.
By component, the Europe medical exoskeleton market is bifurcated into hardware and software. The hardware segment held a larger share of the Europe medical exoskeleton market share in 2023.
In terms of type, the Europe medical exoskeleton market is bifurcated into powered exoskeleton and passive exoskeleton. The powered exoskeleton segment held a larger share of the Europe medical exoskeleton market share in 2023.
Based on extremity, the Europe medical exoskeleton market is segmented into lower body exoskeleton, upper body exoskeleton, and full body exoskeleton. The lower body exoskeleton segment held the largest share of the Europe medical exoskeleton market share in 2023.
By application, the Europe medical exoskeleton market is segmented into spinal cord injury, multiple sclerosis, stroke, cerebral palsy, Parkinson’s Diseases, and others. The spinal cord injury segment held the largest share of the Europe medical exoskeleton market share in 2023.
In terms of mobility, the Europe medical exoskeleton market is bifurcated into mobile exoskeleton and stationary exoskeleton. The powered exoskeleton segment held a larger share of the Europe medical exoskeleton market share in 2023.
By end users, the Europe medical exoskeleton market is segmented into rehabilitation centers, physiotherapy centers, long term care centers, homecare settings, and others. The rehabilitation centers segment held the largest share of the Europe medical exoskeleton market share in 2023.
Based on country, the Europe medical exoskeleton market is segmented into Germany, France, the UK, Italy, Spain, and the Rest of Europe. Germany segment held the largest share of Europe medical exoskeleton market in 2023.
BIONIK; B-Temia Inc; Cyberdyne Inc; Ekso Bionics Holdings Inc; ExoAtlet; Hocoma AG; Lifeward, Inc; Myomo Inc; Rex Bionics Ltd.; and Wearable Robotics SR are some of the leading companies operating in the Europe medical exoskeleton market.
The Europe Medical Exoskeleton Market is valued at US$ 107.48 Million in 2023, it is projected to reach US$ 853.96 Million by 2031.
As per our report Europe Medical Exoskeleton Market, the market size is valued at US$ 107.48 Million in 2023, projecting it to reach US$ 853.96 Million by 2031. This translates to a CAGR of approximately 29.6% during the forecast period.
The Europe Medical Exoskeleton Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Europe Medical Exoskeleton Market report:
The Europe Medical Exoskeleton Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Europe Medical Exoskeleton Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Europe Medical Exoskeleton Market value chain can benefit from the information contained in a comprehensive market report.