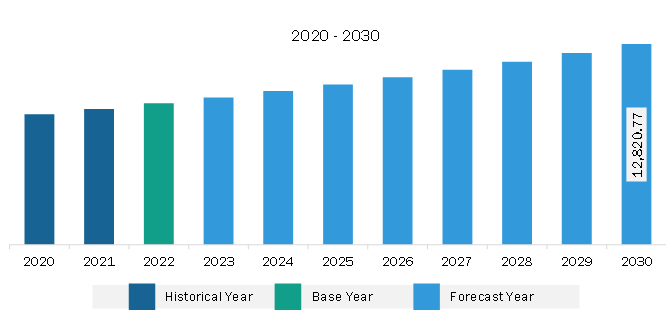
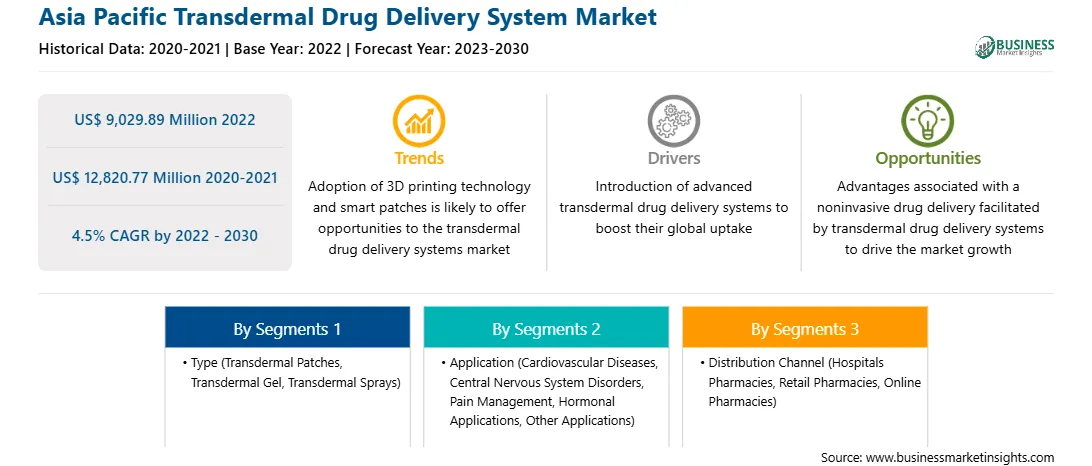
The Asia Pacific transdermal drug delivery system market is expected to grow from US$ 9,029.89 million in 2022 to US$ 12,820.77 million by 2030. It is estimated to register a CAGR of 4.5% from 2022 to 2030.
A transdermal drug administration route is novel and one of the reliable means of sustained drug delivery. With significant research being conducted in this field and the increasing interest of researchers in this form of drug delivery, a large number of transdermal patches are likely to reach the marketplace in the future. Transdermal patches are used to deliver drugs such as nicotine, fentanyl, nitroglycerin, and clonidine to treat various medical conditions. Recently, these patches have been explored to deliver biologics in various applications. Smart patches are equipped with sensors and other technologies to monitor patient conditions and adjust drug delivery accordingly. In January 2023, a lead scientist at Swansea University developed a new smart patch to detect proinflammatory biomarkers of neurodegenerative diseases such as Parkinson's and Alzheimer's using the microneedle technology. These smart patches comprise numerous microneedles designed to penetrate the skin barrier in a minimally invasive manner, followed by monitoring the biomarkers of clinical significance. They can be self-administered for point-of-care diagnosis at general practices or at home. This innovative research can potentially change the landscape of early neurodegenerative disease detection.
According to an article titled "3D-Printed Products for Topical Skin Applications: From Personalized Dressings to Drug Delivery," published in the National Library of Medicine in November 2021, personalized 3D-printed patches that conform to the skin surface can be used for acetyl-hexapeptide 3 (AHP-3) delivery. In this study, two liquid monomers-polyethylene glycol diacrylate (PEGDA) and vinylpyrrolidone (VP)-were used in different proportions to improve the mechanical strength, polymerization rate, and swelling rate of the final polymer. Optimal drug loading on the resin indicated that AHP-3 remained stable throughout the manufacturing process and had no effect on the physical properties of the final polymer. Using a 3D scanned facial model, a personalized patch was designed in computer-aided design software, and it was fabricated in optimized resin using a digital light processing 3D printer. The in-vitro characterization of prepared transdermal patches has shown their ability to penetrate human cadaver skin, further remaining intact upon compression. "3D-Printed Products for Topical Skin Applications: From Personalized Dressings to Drug Delivery" is the first study demonstrating personalized patches made using photopolymers, and it has introduced a novel approach to improve the transdermal delivery of drugs for effective wrinkle management.
Thus, the adoption of 3D printing technology and smart patches is likely to offer opportunities to the transdermal drug delivery systems market in the coming years.
The Asia Pacific transdermal drug delivery systems market is segmented into China, Japan, India, Australia, South Korea, and the Rest of Asia Pacific. The market growth is expected to be fastest in countries such as China, India, and Australia due to growing aging population, increasing prevalence of cardiovascular diseases, and increasing demand for non-invasive medical devices. In Asia Pacific, China is the largest market of transdermal drug delivery systems. The growth of the transdermal drug delivery system market in China is primarily attributed to the growing technology in the country, increasing developments by the market players to develop transdermal patches and semisolid products for pain management.
Also, in the last few years, companies in China are undergoing product launches and approvals, acquisitions, partnerships, and mergers to expand their business in the transdermal drug delivery systems industry. For instance, in January 2022, Suzhou Reveda Medical Co., Ltd. (REVEDA) announced that it partnered with Hybio Pharmaceutical Co., Ltd. (Hybio Pharmaceutical) to set up a joint venture for drug-device combination products and team up in the field of polypeptide micro-needles for transdermal delivery. 60% of the joint venture will be owned by REVEDA Medical and 40% by Hybio Pharmaceutical. Furthermore, in September 2021, the UK's Medicines and Healthcare Products Regulatory Agency granted marketing approval for Luye Pharma Group's innovative formulation Rivastigmine Multi-Day Transdermal Patch (Rivastigmine MD) for the treatment of mild to moderate dementia associated with Alzheimer's disease. Several European Union nations already have marketing authorization for the medicine. Thus, owing to the abovementioned factors, the market is expected to propel the demand for transdermal drug delivery systems in this country in the upcoming years.
Strategic insights for the Asia Pacific Transdermal Drug Delivery System provides data-driven analysis of the industry landscape, including current trends, key players, and regional nuances. These insights offer actionable recommendations, enabling readers to differentiate themselves from competitors by identifying untapped segments or developing unique value propositions. Leveraging data analytics, these insights help industry players anticipate the market shifts, whether investors, manufacturers, or other stakeholders. A future-oriented perspective is essential, helping stakeholders anticipate market shifts and position themselves for long-term success in this dynamic region. Ultimately, effective strategic insights empower readers to make informed decisions that drive profitability and achieve their business objectives within the market.
| Report Attribute | Details |
|---|---|
| Market size in 2022 | US$ 9,029.89 Million |
| Market Size by 2030 | US$ 12,820.77 Million |
| Global CAGR (2022 - 2030) | 4.5% |
| Historical Data | 2020-2021 |
| Forecast period | 2023-2030 |
| Segments Covered |
By Type
|
| Regions and Countries Covered | Asia-Pacific
|
| Market leaders and key company profiles |
The geographic scope of the Asia Pacific Transdermal Drug Delivery System refers to the specific areas in which a business operates and competes. Understanding local distinctions, such as diverse consumer preferences (e.g., demand for specific plug types or battery backup durations), varying economic conditions, and regulatory environments, is crucial for tailoring strategies to specific markets. Businesses can expand their reach by identifying underserved areas or adapting their offerings to meet local demands. A clear market focus allows for more effective resource allocation, targeted marketing campaigns, and better positioning against local competitors, ultimately driving growth in those targeted areas.
The Asia Pacific transdermal drug delivery system market is segmented into type, application, distribution channel, and country.
Based on type, the Asia Pacific transdermal drug delivery system market is segmented into transdermal patches, transdermal gel, transdermal sprays, and others. The transdermal gel segment held the largest share of the Asia Pacific transdermal drug delivery system market in 2022. The transdermal patches is further sub segmented into drug-in-adhesive patches, matrix patches, reservoir membrane patches, and vapor patches.
In terms of application, the Asia Pacific transdermal drug delivery system market is categorized into cardiovascular diseases, central nervous system disorders, pain management, hormonal applications, and other applications. The pain management segment held the largest share of the Asia Pacific transdermal drug delivery system market in 2022. The pain management is further sub segmented into opioid and non-opioid. The hormonal applications is further sub segmented into transdermal estrogen & progesterone therapy and testosterone replacement.
By distribution channel, the Asia Pacific transdermal drug delivery system market is segmented into hospitals pharmacies, retail pharmacies, online pharmacies, and others. The hospitals pharmacies segment held the largest share of the Asia Pacific transdermal drug delivery system market in 2022.
Based on country, the Asia Pacific transdermal drug delivery system market is categorized into China, Japan, India, Australia, South Korea, and the Rest of Asia Pacific. China dominated the Asia Pacific transdermal drug delivery system market in 2022.
Boehringer Ingelheim International GmbH, GSK Plc, Hisamitsu Pharmaceutical Co Inc, Johnson & Johnson, Luye Pharma Group Ltd, Novartis AG, and Viatris Inc are some of the leading companies operating in the Asia Pacific transdermal drug delivery system market.
1. Boehringer Ingelheim International GmbH
2. GSK Plc
3. Hisamitsu Pharmaceutical Co Inc
4. Johnson & Johnson
5. Luye Pharma Group Ltd
6. Novartis AG
7. Viatris Inc
The Asia Pacific Transdermal Drug Delivery System Market is valued at US$ 9,029.89 Million in 2022, it is projected to reach US$ 12,820.77 Million by 2030.
As per our report Asia Pacific Transdermal Drug Delivery System Market, the market size is valued at US$ 9,029.89 Million in 2022, projecting it to reach US$ 12,820.77 Million by 2030. This translates to a CAGR of approximately 4.5% during the forecast period.
The Asia Pacific Transdermal Drug Delivery System Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Asia Pacific Transdermal Drug Delivery System Market report:
The Asia Pacific Transdermal Drug Delivery System Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Asia Pacific Transdermal Drug Delivery System Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Asia Pacific Transdermal Drug Delivery System Market value chain can benefit from the information contained in a comprehensive market report.