The 2019 novel coronavirus (2019-nCoV) was officially named COVID-19 by the WHO that has spread to more than 180 countries, including China; therefore, the disease was named the global pandemic. The spread of the COVID-19 pandemic has emphasized health data sharing and interoperability under the microscope. Pediatric coronavirus disease-19 (COVID-19) infection is comparatively mild compared to adults, and children are assigned a better prognosis. The interruptions in the supply chain and the massive demand for efficient treatments for the therapy of COVID-19 have put the health research industry in the Asia-Pacific region in a crucial position. The COVID-19 pandemic has had a major influence on international trade and commerce. India, too, has suffered owing to unexpected economic disruptions caused by interim restrictions on public activities and regional lockdowns. Asia’s value chains have closely integrated the economies of China, Japan, South Korea, Hong Kong, and other Asian countries. Such dense and extremely coordinated value chains enable exploiting massive economies of scale in manufacturing. China’s manufacturing Purchasing Managers Index (PMI) declined sharply to levels equivalent to those throughout the 2009 international monetary crisis and despite the March rebound, there were many proofs indicating a pointy contraction in growth within the first quarter of 2020. During the COVID-19 pandemic, most programs in this region were affected, but to varying degrees, influenced mainly by the severity of the local pandemic. Medical device manufacturers and suppliers are shifting their focus to developing and supplying COVID-19 essentials and medical devices for respiratory diseases. This scenario harms the market. This pandemic is affecting the operations of the various key players operating in this region. Considering the geographic and economic operations between Asian countries and China, the countries are expected to witness challenge of COVID-19. Domestic start-ups backed by international funding have already started witnessing back out from investors putting the development of the products in jeopardy. In these unprecedented times, biotechnology and pharmaceutical companies are responding to the rapid challenges arising from disruption in supply chains and the need to change business processes. If this pandemic lasts for a longer time, it may impact the supply of active material and ingredients along with the import and export of biotechnology and biopharmaceuticals. There is also the potential for negative impacts of both a medium- and longer-term nature on R&D and manufacturing activities, and delay on projects. These factors are affecting the growth of the E. coli water testing market in the region.
With the new features and technologies, vendors can attract new customers and expand their footprints in emerging markets. This factor is likely to drive the E.Coli water testing kits market. The APAC E.Coli water testing kits is expected to grow at a good CAGR during the forecast period.
Strategic insights for the Asia Pacific E.Coli Water Testing Kits provides data-driven analysis of the industry landscape, including current trends, key players, and regional nuances. These insights offer actionable recommendations, enabling readers to differentiate themselves from competitors by identifying untapped segments or developing unique value propositions. Leveraging data analytics, these insights help industry players anticipate the market shifts, whether investors, manufacturers, or other stakeholders. A future-oriented perspective is essential, helping stakeholders anticipate market shifts and position themselves for long-term success in this dynamic region. Ultimately, effective strategic insights empower readers to make informed decisions that drive profitability and achieve their business objectives within the market.
| Report Attribute | Details |
|---|---|
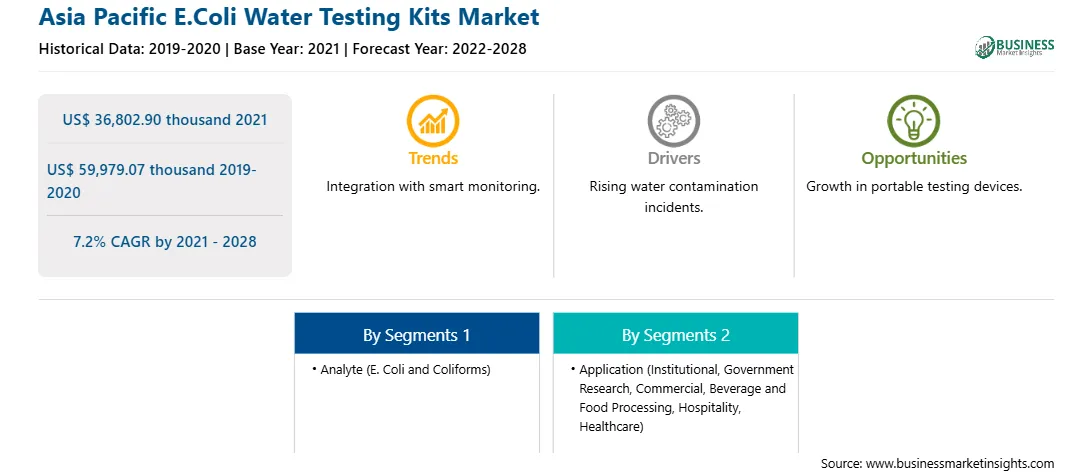
| Market size in 2021 | US$ 36,802.90 thousand |
| Market Size by 2028 | US$ 59,979.07 thousand |
| Global CAGR (2021 - 2028) | 7.2% |
| Historical Data | 2019-2020 |
| Forecast period | 2022-2028 |
| Segments Covered |
By Analyte
|
| Regions and Countries Covered | Asia-Pacific
|
| Market leaders and key company profiles |
The geographic scope of the Asia Pacific E.Coli Water Testing Kits refers to the specific areas in which a business operates and competes. Understanding local distinctions, such as diverse consumer preferences (e.g., demand for specific plug types or battery backup durations), varying economic conditions, and regulatory environments, is crucial for tailoring strategies to specific markets. Businesses can expand their reach by identifying underserved areas or adapting their offerings to meet local demands. A clear market focus allows for more effective resource allocation, targeted marketing campaigns, and better positioning against local competitors, ultimately driving growth in those targeted areas.
The Asia Pacific E.Coli Water Testing Kits Market is valued at US$ 36,802.90 thousand in 2021, it is projected to reach US$ 59,979.07 thousand by 2028.
As per our report Asia Pacific E.Coli Water Testing Kits Market, the market size is valued at US$ 36,802.90 thousand in 2021, projecting it to reach US$ 59,979.07 thousand by 2028. This translates to a CAGR of approximately 7.2% during the forecast period.
The Asia Pacific E.Coli Water Testing Kits Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Asia Pacific E.Coli Water Testing Kits Market report:
The Asia Pacific E.Coli Water Testing Kits Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Asia Pacific E.Coli Water Testing Kits Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Asia Pacific E.Coli Water Testing Kits Market value chain can benefit from the information contained in a comprehensive market report.